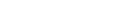Water Based Liquid Scintillator555
Why do we need it?
Water-based Liquid Scintillator (WbLS) would be extremely useful for experiments that need very large mass neutrino targets (50,000 tons or more) where use of conventional organic Liquid Scintillator (LS) is expensive and difficult to deal with from an environmental standpoint. It is also useful in that many isotopes can be dissolved in water that are fairly insoluable in the non-polar solvents used for conventional LS. WbLS is being developed for potential use in experiments like ANNIE, WATCHMAN, and THEIA.
How does it work?
Conventional scintillator typically works by exciting a benzene ring in an organic molecule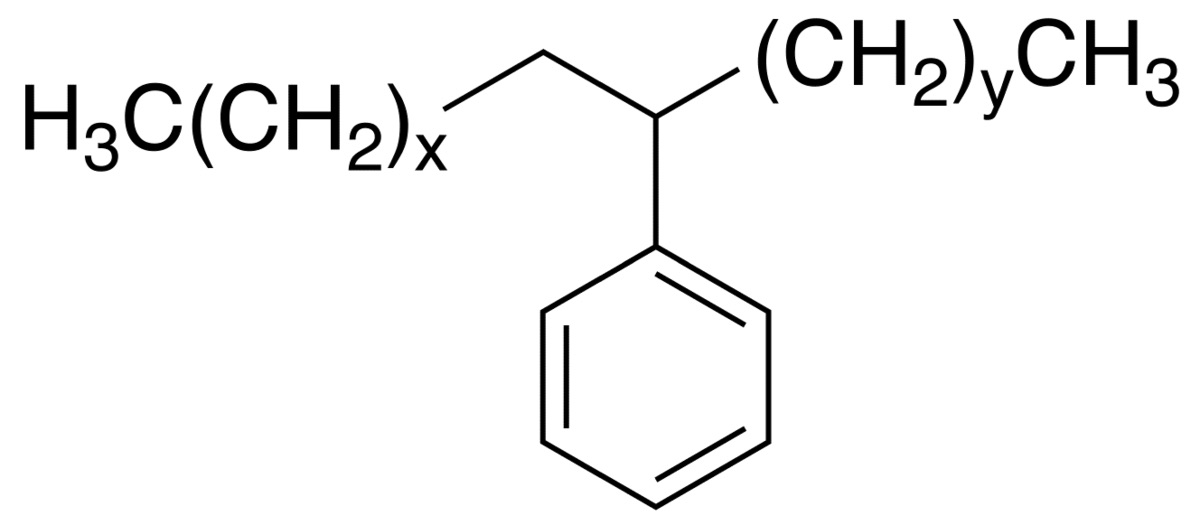 (such as Linear Alkylbenzene - LAB, as shown on the right) via ionization energy transfer from a charged particle passing nearby. The ring then de-excited by emitting a photon, which then is absorbed by a wave shifting fluor, such as 1,4-Bis(2-methylstyryl)benzene (Bis-MSB). The wave shifter avoids resonant self-absorption by shifting the spectrum to an optical region where the organic liquid is more transparent. Unfortunately, such organic liquids are almost always not miscible in water.
(such as Linear Alkylbenzene - LAB, as shown on the right) via ionization energy transfer from a charged particle passing nearby. The ring then de-excited by emitting a photon, which then is absorbed by a wave shifting fluor, such as 1,4-Bis(2-methylstyryl)benzene (Bis-MSB). The wave shifter avoids resonant self-absorption by shifting the spectrum to an optical region where the organic liquid is more transparent. Unfortunately, such organic liquids are almost always not miscible in water.
WbLS gets around this by encapsulating the organic liquid in tiny (100 nanometer scale)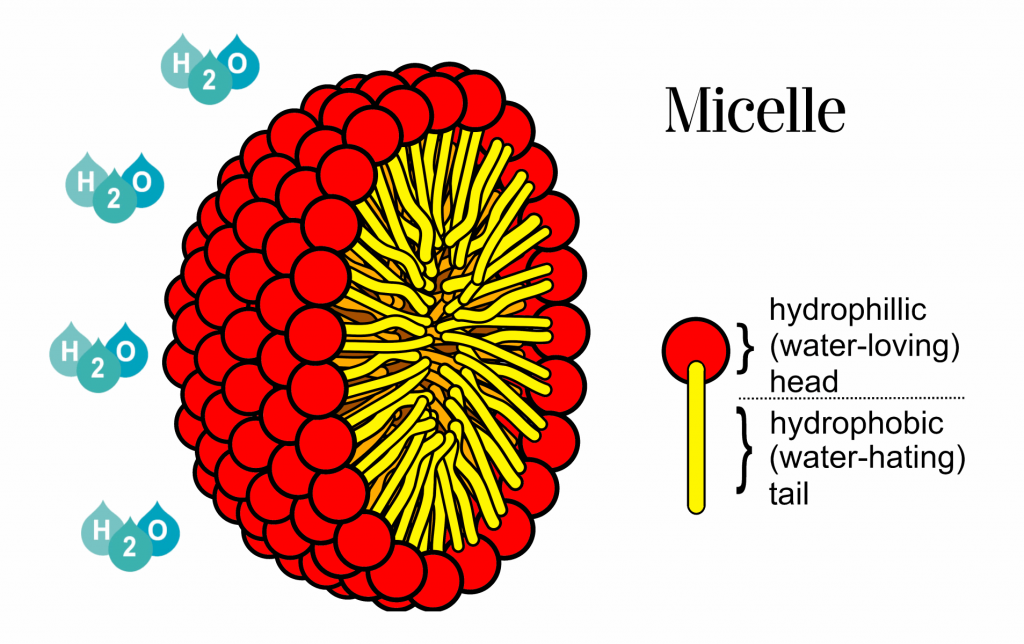 micelles that are formed by a class of chemicals called surfactants. A surfactant is a chain molecule that has a chemically hydrophillic end and a hydrophobic end. The hydrophobic end will escape being in water by burying itself in the organic liquid. The hydrophillic ends will happily terminate in water. A generic micelle is shown in the picture on the right. The organic LS is then confined inside the micelle and can be mixed into water in the desired concentration.
micelles that are formed by a class of chemicals called surfactants. A surfactant is a chain molecule that has a chemically hydrophillic end and a hydrophobic end. The hydrophobic end will escape being in water by burying itself in the organic liquid. The hydrophillic ends will happily terminate in water. A generic micelle is shown in the picture on the right. The organic LS is then confined inside the micelle and can be mixed into water in the desired concentration.
Testing WbLS at UC Davis
Developed at Brookhaven National Lab, we are working on two important aspects of  making WbLS a practical option for neutrino experiments: (1) care must be taken than detector components are compatible with the chemically active WbLS, and (2) there must be a way of purifying WbLS in situ in order to remove light absorbers leeching into the water target from plasticizers and/or exposed metals (like stainless steel). UC Davis has a Nanofiltration (NF) laboratory, complete with a test bench NF system, Shimadzu photo spectrometer, and Neutron Activation Analysis tracer capability. The picture below shows UC Davis graduate student Morgan Askins happily making WbLS in the chemistry lab at Brookhaven.
making WbLS a practical option for neutrino experiments: (1) care must be taken than detector components are compatible with the chemically active WbLS, and (2) there must be a way of purifying WbLS in situ in order to remove light absorbers leeching into the water target from plasticizers and/or exposed metals (like stainless steel). UC Davis has a Nanofiltration (NF) laboratory, complete with a test bench NF system, Shimadzu photo spectrometer, and Neutron Activation Analysis tracer capability. The picture below shows UC Davis graduate student Morgan Askins happily making WbLS in the chemistry lab at Brookhaven.